Home »

Nearly 200 delegates from nine countries came together for this year’s Type 1 Diabetes Grand Challenge Symposium. A gathering that not only provided updates on the world-leading science funded by the programme but also demonstrated just how much the Type 1 Diabetes Grand Challenge community has grown in just over three years.
If last year’s event was filled with anticipation and ambitious possibility, this year’s was defined by confidence, progress, and palpable momentum. Across every session, the message was clear: The Grand Challenge is delivering impact, and the pace is accelerating.
Turning discovery into impact
We heard about the progress of research exploring new immunotherapy regimens, beta cell replacement and regeneration techniques, and innovative strategies to protect and support transplanted beta cells.
Speakers highlighted how scientific advances made over the past twelve months have shifted the conversation. Where last year’s discussions carried a tone of speculation, this year was marked by tangible achievement: new breakthroughs, new collaborations, and critically, the Grand Challenge’s first in-person trials now underway, with more moving toward the clinic.
Dr Elizabeth Robertson, Director of Research at Diabetes UK, captured this sense of progress:
“It’s an incredibly exciting time for beta cell therapy and immunotherapy research, as we work toward replacing lost beta cells and protecting them from immune destruction in people with type 1 diabetes. To move closer to real-world treatments, we’re partnering not only with world-class researchers but also with experts in translational medicine – the people who can speed up the journey from lab discovery to clinical reality. These are complex scientific challenges and solving them will require the very best minds working together, collaborating openly, and sharing both successes and setbacks. You never know which discovery will become the critical piece of the puzzle that opens the door to new type 1 treatments.”
A community working differently together
The Grand Challenge was founded on the belief that collaboration is essential to accelerating progress. At this year’s symposium, that belief was made real.
Every talk, panel, and poster demonstrated how teams across disciplines, continents, and career stages are working as a unified community, from immunologists and bioengineers to data scientists and experts in clinical translation.
Seeing those collaborations in person brought the programme’s design to life. As Liam Eaglestone, CEO of the Steve Morgan Foundation, noted:
“We’ve created a community that is making a difference. What was a risky hope three and a half years ago is starting to deliver. This will only happen if we bring all the different partnerships and expertise together to achieve something remarkable.”
Steve Bates, Trustee at Breakthrough T1D, echoed this sentiment:
“The Grand Challenge is a unique partnership between the Steve Morgan Foundation, Breakthrough T1D and Diabetes UK. We’re especially grateful for all the time that so many leading type 1 investigators, clinicians and experts by experience are giving to help us drive progress. Bringing together groups under a common goal is key to driving real change fast. I know the Grand Challenge will help drive research all the way through to commercialisation, delivering new therapies and cures for people with type 1.”
Putting lived experience at the centre
Another theme running through the symposium was the prominent role of experts by experience in every aspect of Grand Challenge-funded research – from shaping research priorities and contributing to funding decisions, to supporting the design, delivery and dissemination of research projects.
Every speaker, from early-career researchers to senior investigators, acknowledged how lived experience is shaping their research. Delegates heard directly from adults living with type 1, parents of children with the condition and members of the Grand Challenge’s public and patient involvement (PPI) groups.
Their insights are not an ‘add-on’ to the science: they are fundamental to it, helping researchers think deeply about the issues they are trying to solve and putting people affected by type 1 at the heart of every decision.
Looking ahead
With multiple research studies now progressing and a research community more connected than ever, the Grand Challenge is entering an exciting new phase.
What began as a bold, high-risk, high-reward vision is becoming a programme set to deliver real-world impact. It’s driven by collaboration, guided by lived experience, and fuelled by scientific excellence.
And if the energy in the room at this year’s symposium was anything to go by, the pace of progress is only set to increase.
A lifetime shaped by research, now shaping research
November 14, 2025
Sarah Gatward’s earliest experiences with type 1 diabetes have sparked a lifetime of curiosity and involvement in life-changing research. More than 50 years on, she’s helping others to get involved, to make the Type 1 Diabetes Grand Challenge as impactful as possible for people living with type 1.
Sarah was diagnosed with type 1 diabetes aged 7, after being rushed to hospital, semi-conscious, on 5 March 1972. She was in and out of hospital for a month.
“There were very regimented visiting hours and when the consultant doctors were doing ward rounds parents had to wait outside. The hospital team didn’t want to speak to them.”
Frustrated by this wall of silence, and worried sick about her young daughter, Sarah’s mother urged her to listen carefully to what was being said about her condition, so she could pass it on to her parents and help to look after herself.
“My parents believed very strongly that I needed to understand and be involved with it right from the start. I think it’s helped me enormously.”
A formative experience of research
This early curiosity meant that when an opportunity came up to take part in research into insulin pumps while the family was living in the US, 15-year-old Sarah was keen.
“I’d hit adolescence and everything changed enormously. I was having bad hypoglycaemia overnight which would lead to nighttime convulsions. There was no easy way of trying to work out what was causing it and how [my doctors] should manage it”
Sarah joined a one-year research trial, which showed that insulin pumps achieved better blood sugar management than three insulin injections per day in young people. This was an important finding for the future of type 1 treatment and had a life-changing influence on how Sarah managed her condition.
“Taking part in the pump research provided me with the means to adjust my treatment to suit my day-to-day life. Until that point, I’d had quarterly hospital reviews where dose adjustments were made before I was sent home to continue with that fixed pattern for the next three months. I had learned it worked better to flex my treatment according to daily needs, and the pump allowed me to do that.”
Getting involved
That first encounter with research was just the start. Over the years Sarah has participated in other clinical trials looking at different aspects of type 1 management, before getting involved as a patient adviser.
“Breakthrough T1D were looking for a group to review and approve small grant award applications. It was made up of healthcare professionals, researchers and patients. I brought my experience of life with type 1 and professionals brought their knowledge of the condition and we worked together. I sat on the committee for four years and it was a really positive experience.”
Lived experience in the Grand Challenge
During her time on the Scientific Advisory Board, Sarah got to know several researchers working in the field. This led to her being invited by Professor Shanta Persaud and Professor Aileen King to assist with an application for an ambitious research application to the Grand Challenge.
Their beta cell therapy programme was funded, and Sarah now leads a group of people with lived experience of type 1 who work with the research team. This is commonly known as Patient and Public Involvement (PPI) and—as Professor King explains—the Grand Challenge ensures PPI isn’t just a box-ticking exercise.
“We have really benefited from having Sarah as a co-applicant on our grant. While we’ve engaged with patients in the past, it was never as structured or sustained as it has been through the Grand Challenge. With Sarah, we’ve built a PPI group that we’re really proud of. We hold regular meetings and are planning the group’s second visit to our labs. It’s been a privilege to get to know them.”
The PPI group brings valuable first-hand insights from people of different ages and experiences. For instance, two members have had islet cell transplantation. They shared their insights in a session for the researchers, who are considering the practicalities of delivering stem cell-derived beta cells to people with type 1 diabetes.
Professor King continues:
“It was really eye-opening for us. Although we were aware of the scientific practicalities of the procedure, it brought to light the process from a patient point of view. We gained a deeper understanding of what it’s truly like to wait for a transplant, and the significant sacrifices patients often have to make afterwards.
“I think the whole team felt really inspired. Sometimes science is hard and bringing extra inspiration to our team to remind them why they are doing these difficult experiments is important. These insights [also] remind us to stay mindful of what patients actually want to achieve. It helps guide our research to remain aligned with their needs and expectations.”

Researchers at King’s College London with their Patient and Public Involvement members
Optimism and hope
Sarah describes herself as a ‘realistic optimist’; believing that research can improve things but knowing that it can take time. This outlook has been shaped by the incredible advances in type 1 treatment she’s experience over the decades, which were unimaginable to her as a child.
“I had the kit I needed for urine testing in our downstairs toilet and it was all quite squished balancing the equipment around the edge of the wash basin. I remember fantasising that when I’m an adult I’m going to have a special room that I can have all my medical kit in so that I could spread it out and have space around me. It just never occurred to me that I would no longer be undertaking urine tests and that the equipment available for managing the condition would be so much more advanced!”
Now, with the Grand Challenge, Sarah’s sights are set on a day when she doesn’t have to be injecting insulin at all.
“We’re getting closer and closer. I’ve talked about it with a couple of friends who have also been living with type 1 long term and we’ve often joked about what on earth we would do with all the spare mental capacity that a cure would bring! It would be absolutely wonderful to have that headspace back, having a life where I don’t have to think about type 1 all the time.”
New clues to why type 1 diabetes is more aggressive in young children
November 13, 2025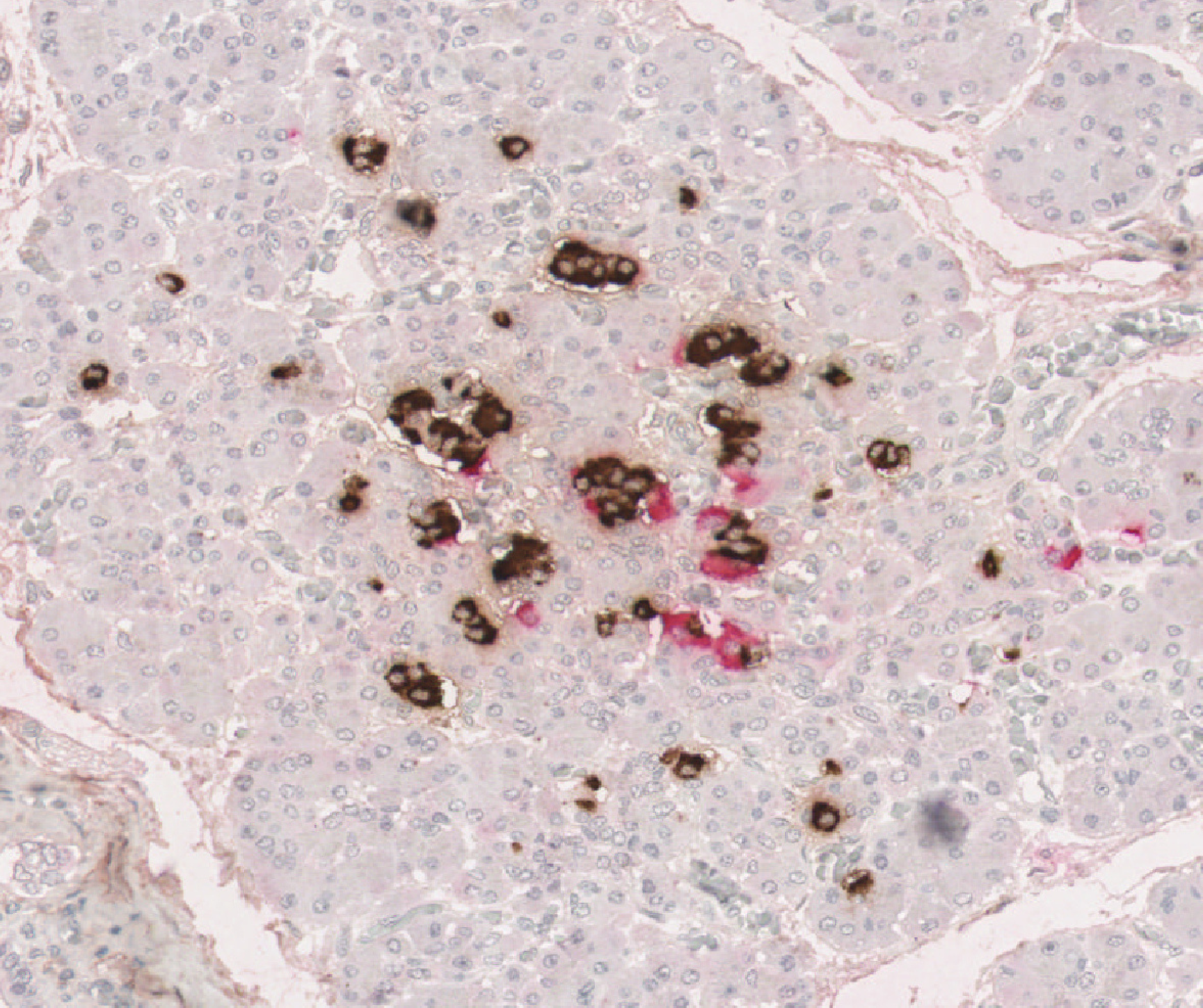
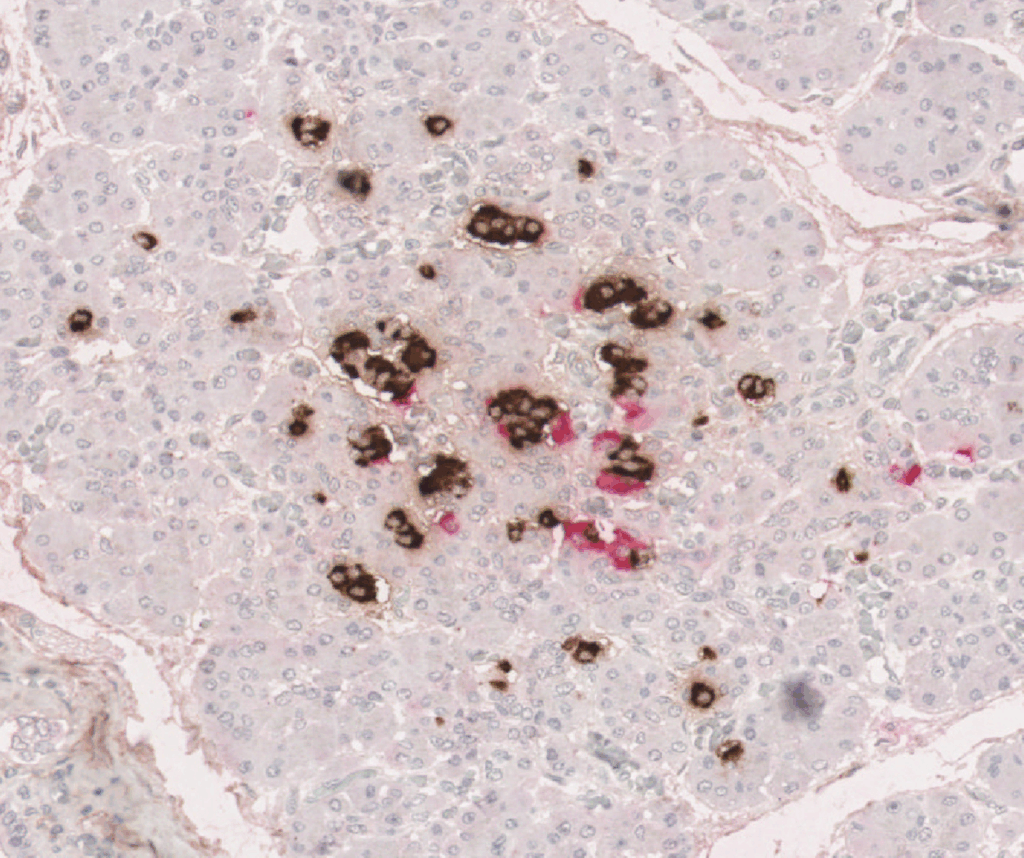
Professor Sarah Richardson and her team have made a major breakthrough in understanding why type 1 diabetes is more aggressive in young children, revealing that nearly all their insulin-producing beta cells are destroyed before they can mature. The new insights could pave the way for new strategies to prevent or delay type 1 diabetes and, in time, contribute to a cure.

Professor Sarah Richardson, looking at islet samples through a microscope in her lab.
We know that type 1 diabetes is an autoimmune condition, where the immune system attacks and destroys the beta cells in the pancreas. In young children, typically under the age of 7, the immune attack and its destruction of beta cells typically progress rapidly. This can increase the likelihood of diabetic ketoacidosis at diagnosis and make the condition particularly difficult to manage.
Until now, scientists had limited tools to study the early development of beta cells, which are found in clusters in the pancreas. In young children, these clusters are small and still forming, and only contain a few beta cells.
In their latest study published in Science Advances, Professor Richardson and her team used cutting-edge scientific techniques to study these small clusters in unprecedented detail. They analysed rare pancreas samples from over 250 people of varying ages, both with and without type 1 diabetes. They looked at how these clusters change as we age and how they are affected by the immune system.

Teifion Luckett from Sarah’s team, looking at data for their research
The findings confirm that in early childhood, people without type 1 diabetes have many small clusters of beta cells, which normally increase in size and mature with age, with the most rapid development occurring in the first few years of life.
For the first time, our Grand Challenge researchers showed that in people with type 1 diabetes these small clusters are almost completely absent, having been destroyed by the immune system. While some people with type 1 diabetes retained a few large clusters, allowing them to produce small amounts of their own insulin, this was not the case for those diagnosed at a young age.
Together, the results suggest that the abundant small clusters found in young children are especially vulnerable to the type 1 diabetes immune attack. Their rapid destruction prevents them from maturing, leaving very few beta cells later in life. This explains why children diagnosed with type 1 diabetes at a young age typically are unable to produce any of their own insulin, making the condition particularly difficult to manage.
This research underscores the critical role of these small clusters in healthy pancreas development and opens the door for new treatments to protect children’s small clusters of beta cells, giving them the chance to mature into large clusters that are less vulnerable to the immune attack. It also strengthens the case for early type 1 diabetes screening – particularly in young children – essential for identifying those in the early stages of type 1 diabetes before these crucial cells are lost.
Professor Sarah Richardson said:
“These tiny insulin-producing beta cell clusters – once overlooked – hold big clues to understanding type 1 diabetes. This new perspective has the potential to reshape how we screen, treat, and even prevent type 1 diabetes. Protecting small beta cell clusters early could be key to stopping type 1 diabetes before it starts.”
Gareth and Joanne Nye’s daughter was diagnosed with type 1 diabetes at just 23 months old. They told us:
“Gracie’s diagnosis was traumatic for our whole family. In less than 48 hours she went from being a toddler with what we thought was a slight cold, to lying unconscious in a hospital bed with diabetic ketoacidosis (DKA), close to death. We lived in constant fear, setting alarms every two hours to finger prick her at night, worrying if she’d still be with us in the morning.
“Research like this, and the possibilities it holds, will be vital in reducing the number of children diagnosed in critical care, like Gracie. It gives us confidence that one day she could be free from her condition – and that fewer parents and children will have to go through this same experience.”
Dr Elizabeth Robertson, Director of Research and Clinical at Diabetes UK, said:
“The Type 1 Diabetes Grand Challenge set out to fund bold, ambitious research with the potential to fundamentally shift progress toward new treatments and, ultimately, a cure for type 1 diabetes. This study delivers on that vision by challenging the foundations of previous understanding about the development of type 1 diabetes in early childhood.
“Uncovering why type 1 diabetes is so aggressive in young children opens the door to developing new immunotherapies aimed at slowing or stopping the immune attack, potentially giving children more precious years without insulin therapy and, one day, preventing the need for it entirely.”
Rachel Connor, Director of Research Partnerships at Breakthrough T1D, said:
“This study gives us a missing piece of the puzzle, explaining why type 1 diabetes progresses so much faster in children than in adults. For families, that rapid progression can turn everyday life upside down, with a child becoming seriously unwell before the condition is even recognised and parents having to take on a demanding new routine overnight. By revealing how the condition behaves differently in young children, these insights can guide the development of more effective, targeted treatments for young people living with type 1 diabetes.”
Early research on “smart” insulin brings hope for safer type 1 diabetes management
October 6, 2025
Researchers funded by the Type 1 Diabetes Grand Challenge have developed a new insulin–glucagon molecule, which could reduce dangerous drops in blood glucose, known as hypoglycaemia. For people living with type 1 diabetes, where hypoglycaemia is a constant risk, this breakthrough has the potential to make daily management safer and more reliable.
In type 1 diabetes (T1D), a person’s body doesn’t produce enough insulin, meaning blood glucose levels will continue to climb if insulin is not given (either by injection or using a pump). Managing blood glucose levels is a constant balancing act for people with type 1 diabetes, between administering the correct amount of insulin and their blood glucose going too high or too low and dealing with the consequences of this. If blood glucose goes too high, this can cause a potentially fatal complication called diabetic ketoacidosis, where a person’s blood becomes too acidic. Sustained high blood glucose levels can damage the eyes, feet, heart and kidneys. If blood glucose levels drop dangerously low it can lead to seizures, coma, and can sometimes be fatal.
Findings by US-based researchers, published in ACS Pharmacology and Translational Science, show success in engineering a product that combines both glucagon and insulin in the same molecule. Insulin is the hormone responsible for reducing blood sugars by enabling cells to use glucose for energy, and glucagon is the hormone responsible for a raising in blood glucose. This molecule was then able to take advantage of the body’s built in ‘on/off switch’ in the liver. The liver naturally responds more to insulin when glucose is high and more to glucagon when glucose is low. When blood glucose is high, the insulin part of the molecule is active, lowering blood glucose like regular insulin. However, when blood glucose is low, the glucagon part is active, signalling to the liver to release glucose and preventing low blood sugar episodes (known as hypoglycaemia or hypo).
The research team tested the new insulin in rats, with positive initial results. The insulin behaved as intended: lowered blood glucose when high and helped raise blood glucose when low – unlike regular insulin. It also reduced the need for emergency glucose injections in the rats during hypoglycaemia.
The two parts of the molecule (insulin and glucagon) worked independently but in balance, just like separate hormones. The promising results indicate that the new molecule could help lower the chance of hypoglycaemia in people with type 1 diabetes.
The study also showed that the new form of insulin stayed stable for weeks without the need for refrigeration before opening, giving it a longer shelf life than expected. This makes it more reliable for people with T1D and easier for manufacturers to produce, store, and transport.
Low blood glucose is as unpleasant side effect of type 1 diabetes. It normally occurs due to an imbalance between insulin dose, food intake and physical activity. Having a hypo is something which, if possible, all people with type 1 diabetes would want to avoid. It can be dangerous as when blood glucose is low as the brain is deprived of its energy source – glucose. This leads to a sensation of weakness, dizziness or blurred vision, alongside feeling angry or confused. There are many other potential symptoms.
This research has been funded by the Type 1 Diabetes Grand Challenge, a partnership between Breakthrough T1D, Diabetes UK and the Steve Morgan Foundation.
Michael Weiss, distinguished Professor in the Department of Biochemistry & Molecular Biology, Indiana University School of Medicine, said:
“For the past century, coping with hypoglycaemia (the lows) has been an ever-present challenge in type 1 diabetes. This has made creating glucose-responsive insulins (smart insulins) a major goal. Our approach simplifies such design by exploiting an endogenous ‘smart’ switch in the liver, how the body naturally adjusts relative hormonal responses based on whether the blood glucose level is high or low: Too high, insulin wins; too low, glucagon wins!”
Michael Weiss was also supported by lab partner and colleague at Yale, Associate Professor Raimund Herzog, who was part of the team proposal and oversaw the animal testing.
Rachel Connor, Director of Research Partnerships at Breakthrough T1D UK, said:
“This new development in novel insulins research hold exciting promise for people with T1D. Avoiding low blood glucose is a constant balancing act for people with type 1 diabetes, as so many factors can combine to affect blood glucose levels. An insulin offering protection from hypoglycaemia could have a profound impact on the mental burden of living with T1D. With funding from the Type 1 Diabetes Grand Challenge, we’re excited to be driving innovations as fast as possible toward testing with people who live with T1D.”
Dr Elizabeth Robertson, Director of Research and Clinical at Diabetes UK, said:
“The Type 1 Diabetes Grand Challenge is committed to supporting innovation that meets the needs of people living with type 1 diabetes. These positive results are an exciting step towards next-generation insulins that could ease the burden of daily management with insulin therapy. We look forward to seeing this promising research advance into human studies and, ultimately, improve the lives of everyone who depends on insulin therapy.”
With new research such as this, there is a possibility to reduce the number of hypos, which would be safer for all people living with type 1 diabetes. This research is in its early stages, with many more steps to occur before it will be made available to the public. The eventual aim is to create two different types of insulin; a longer lasting version for use once a week, and a short acting version for use in insulin pumps.
Funding boost enables UK researchers to access European type 1 diabetes cell therapy collaboration
September 23, 2025
UK researchers will be able to collaborate with European experts on a major funding initiative to develop cell therapies for type 1 diabetes, thanks to funding from the Type 1 Diabetes Grand Challenge in partnership with UK Research and Innovation (UKRI).
Earlier this year, the UK Government confirmed that UK researchers would be unable to access EU funding for four European Innovation Health Initiative (IHI) call topics – part of the Horizon Europe Framework Programme – due to budget timing issues.
This included the high-profile ‘Leveraging Europe’s Expertise to Accelerate Cell Therapy for Type 1 Diabetes’ call topic, which aims to drive the widespread adoption of beta cell therapy by ensuring its efficacy, accessibility, and integration into healthcare systems.
This risked excluding eligible UK researchers from one of the largest collaborations in the field, potentially leading to duplicated efforts across Europe and slowing progress.
Now, the Grand Challenge will commit up to £2 million over 5 years to ensure UK institutions can participate in the IHI call. The funding will be administered through UKRI.
This investment will mean UK researchers can stay fully embedded in European collaboration and strengthen international knowledge-sharing, to accelerate progress towards life-changing new treatments for people living with type 1 diabetes.
Dr Elizabeth Robertson, Director of Research and Clinical at Diabetes UK, said:
“This funding from the Steve Morgan Foundation will help ensure UK researchers can stay at the forefront of beta cell therapies. It reflects the Type 1 Diabetes Grand Challenge’s determination to act with urgency and to safeguard collaborations, so that people with type 1 diabetes can benefit from life-changing therapies without delay.”
Liam Eaglestone, Chief Executive Officer of the Steve Morgan Foundation, said:
“At the Steve Morgan Foundation, we are committed to transforming the lives of people with type 1 diabetes. When it became clear that additional support was needed for UK researchers to join this important European collaboration, we acted quickly with our Type 1 Diabetes Grand Challenge partners to help make that possible. This funding will ensure UK expertise continues to play a central role in international efforts to advance cell therapies, bringing us closer to life-changing treatments for everyone affected by type 1 diabetes.”
Rachel Connor, Director of Research Partnerships at Breakthrough T1D UK, said:
“We believe that curing type 1 diabetes will take a truly global effort. That’s why we’re committed to ensuring UK researchers can contribute to and benefit from international collaborations like this one. By unlocking access to world-class partnerships, we’re accelerating progress towards breakthrough therapies and keeping the UK firmly embedded in the global push for cure.”
Professor Christopher Smith, the International Champion of UKRI and the Executive Chair of the Arts and Humanities Research Council (ARHC), said:
“This funding enables UK researchers to remain engaged in a major European initiative advancing cell therapies for type 1 diabetes. It reflects UKRI’s commitment to international partnerships that accelerate scientific progress and deliver real-world impact. This collaboration will support UK institutions to drive forward transformative treatments that could change lives across the UK and beyond.”
The first-stage application deadline for the call is 9 October.
Further Information
- Please note that: (1) the UK funding will be administered by UKRI in parallel to (but separate from) the UK Horizon Europe Guarantee and will use largely similar rules; and (2) there will be a requirement for UK organisations to submit a short form with basic information to UKRI and Diabetes UK.
- Potential UK applicants (or their collaborators) should contact Jo Frost – the UK National Contact Point (NCP) for Horizon Europe Health – for further details as soon as possible at ncp-health@iuk.ukri.org
- Mirroring the UKRI Horizon Europe Guarantee process, applicants will be bound by: the IHI project’s Consortium Agreement; relevant clauses in the IHI Grant Agreement; and the terms and conditions of the UKRI Horizon Grant Offer Letter.
In addition, for this call and as part of the Diabetes UK funding conditions, applicants will also be required to agree to Diabetes UK’s standard revenue share agreement in relation to future possible use of IP. Applicants are advised to review section 3 of the Diabetes UK Revenue Share Agreement.
We would advise applicants to share the Diabetes UK standard revenue agreement with their international consortium straight away and ensure that the consortium is aware of the content of the agreement. - Further information on IHI Call 11 Topic 4 “Leveraging Europe’s expertise to accelerate cell therapy for type 1 diabetes” is available on the IHI website.
Designing the future of type 1 diabetes treatment with smart insulins
September 10, 2025
Professor Matt Webber, a biomedical engineer, is working to make insulin smarter. Funded by the Type 1 Diabetes Grand Challenge, he is leading the development of next-generation insulins, which respond in real time to blood sugar levels. His research could lead to safer, more precise ways of managing blood sugar levels for people with type 1.
Can you tell us a bit about your background and what led you to become a biomedical engineer?
I started out in chemical engineering because I was fascinated by the idea of designing materials from the molecular level. Over time, I became especially interested in how those materials could interact with the human body. That interest led me to pursue a PhD in Biomedical Engineering, where I focused on chemistry and materials science as tools to develop better biomaterials and drug delivery systems
What inspired you to focus your research on type 1 diabetes and the development of new types of insulin?
Managing type 1 diabetes takes a tremendous amount of daily effort, constant attention, planning, and mental energy. I was drawn to the idea of developing more autonomous therapies that could help ease that burden. For this to work, the system needs to be incredibly precise, delivering just the right dose at just the right time to safely manage blood sugar levels. Designing insulin and glucagon therapies that respond automatically to changes in glucose levels felt like a really exciting and meaningful challenge where our science could have a real impact.
What does a typical day look like for you in the lab?
No two days are exactly the same, but I usually spend time checking in with students, discussing experiments, analysing data, and planning next steps. I’m constantly thinking about how we can improve our technologies, identify remaining challenges, and move closer to clinical use. Each day brings a new mix of science, mentoring, problem-solving, and deadlines and that variety is what makes the work so engaging and rewarding.
You’re developing a ‘smart insulin’ that responds to blood sugar levels. How does this technology work?
You can think of our technology as giving insulin a built-in sensor. It stays mostly ‘off’ when blood sugar is in a healthy range, but turns ‘on’ and begins working as blood sugar levels rise. The big-picture goal is an insulin that responds automatically as needed to keep blood sugar in check. A more practical near-term goal might be a single insulin that can cover both basal and mealtime needs by varying its potency. That way, people can dose more confidently without constantly calculating or worrying about going low.
What are the next steps to bring your smart insulin from the lab to people living with type 1 diabetes?
We’re focused on making the system more reliable and consistent, including testing it under real-world conditions like exercise or missed meals. We’re also working to ensure it is stable, safe, and suitable for repeat dosing. Eventually, we’ll need to collaborate with industry partners to scale up production and with clinical teams to begin human trials. There’s still plenty of work ahead, but we’re making steady progress toward bringing better therapies to people.
What has been the most rewarding moment in your research journey so far?
One of the most rewarding parts of my job is mentoring students and watching them grow into independent scientists. But what motivates me just as much is the hope that our work can lead to real improvements in people’s lives. I often speak with parents of children with type 1 diabetes, and I see how much they’re counting on us to create safer, better therapies. That hope is powerful, yet it comes with a deep responsibility to turn it into something real. That’s what keeps us going
Has anything surprised you about your research so far?
We might design a system with a very specific function in mind, but once it interacts with the body, things don’t always go according to plan. At first that can be frustrating, but more often than not, it opens the door to unexpected insights. Those moments when the biology teaches us something new can be the most fascinating and valuable parts of the process
What keeps you motivated in your work, especially when things get tough?
It’s the challenge that drives us. If this work were easy, it probably wouldn’t be worth doing. People are counting on us to raise the standard of care, and the possibility that what we’re building could help them is all the motivation we need to keep going.
What message would you like to share with families and people living with type 1 diabetes?
We’re driven every day by the goal of delivering better options for you and your loved ones. Our team is working hard to create tools that make managing type 1 diabetes simpler and safer. Until a real and lasting cure is realised, which I hope is not far off, we’ll keep working to improve the treatment options available now.
The Grand Challenge partners with the NIHR to establish a UK-wide Type 1 Diabetes Cell Therapy Clinical Trials Network
August 4, 2025
We’re excited to announce that we’re investing £5 million over the next five years to establish a UK-wide Type 1 Diabetes Cell Therapy Clinical Trials Network, in partnership with the National Institute for Health and Care Research (NIHR).
This bold initiative will secure the UK’s position as a global leader in type 1 diabetes research, and ensure people living with the condition gain access to the latest cutting-edge treatments as early as possible.
With momentum from the Grand Challenge, the pace of innovation in cell therapies for type 1 diabetes is rapidly accelerating. Several groundbreaking treatments are now advancing into clinical trials, offering new hope for transformative cures. Expanding the UK’s capacity and capability to deliver these therapies into clinical practice through a nationally coordinated approach is now critical.
Supported by the NIHR’s clinical research infrastructure to guarantee sustainability and lasting impact, the Network will pinpoint and address the unique challenges of type 1 diabetes cell therapy trials, while enhancing and integrating existing resources. By leveraging expertise, infrastructure, and data from across the UK, it will provide a world-class, globally attractive platform for type 1 diabetes cell therapy clinical trials.
The Network will connect existing type 1 diabetes networks and bring together leaders from across NIHR’s Biomedical Research Centres (BRCs) and Clinical Research Facilities (CRFs). It will also draw on knowledge beyond type 1 diabetes, tapping into cross-disciplinary experience from areas including oncology and organ transplantation.
Initial focus areas for the Network include:
- patient cohorts and phenotyping;
- patient recruitment;
- involvement of people affected by type 1 diabetes;
- clinical trial delivery models.
A Task and Finish Group, chaired by Professor Simon Heller, an internationally recognised diabetes expert and Chair of the Grand Challenge Scientific Advisory Panels, and comprising clinical leaders and people with lived experience of type 1 diabetes, will guide the Network’s strategy and implementation.
To accelerate progress, a senior NIHR expert will be embedded within the Grand Challenge team, and a new funding call will launch to strengthen infrastructure for delivering beta cell therapy trials across the UK. Further details will be shared in late 2025.
Dr Elizabeth Robertson, Director of Research and Clinical at Diabetes UK, said:
“We’re delighted to partner with the NIHR to establish the UK’s first Type 1 Diabetes Cell Therapy Clinical Trials Network, aimed at delivering breakthrough, potentially curative therapies. This initiative captures the very essence of the Grand Challenge: ambitious partnerships, cutting-edge innovation, and an unwavering focus on improving the lives of people with type 1 diabetes as quickly as possible. It’s a strategic leap toward making cell therapies a real-world treatment option, while reinforcing the UK’s position as a global leader in clinical research.”
Professor Marian Knight, Scientific Director for NIHR Infrastructure, said:
“This new clinical trials network will bring together world-leading expertise in type 1 diabetes with the power of the NIHR’s infrastructure to accelerate progress towards the development of cutting-edge cell therapies. Putting patients at the heart of the new research will ensure these new treatments are rapidly transitioned from bench to bedside, improving the lives and futures of people with diabetes.”
Liam Eaglestone, Chief Executive Officer of the Steve Morgan Foundation:
“We’re proud to fund the work of the Type 1 Diabetes Grand Challenge, supported by NIHR. By bringing together the best people and organisations into diabetes research, this new network will speed up the development of life-changing treatments, bringing us closer to a future without type 1 diabetes”
The innovation injection: the cross-disciplinary push to cure type 1 diabetes
July 14, 2025
Humans like comfort zones. We have a tendency to stick to the familiar and spend time with people like ourselves. But this creates blind spots and, in research, those blind spots could contain the next big breakthrough.
The Type 1 Diabetes Grand Challenge is enticing leading experts from across science and engineering to bring fresh perspectives and drive innovations. The aim is to increase the pace of progress and overcome longstanding challenges, so that we find better treatments and a cure for type 1 diabetes as soon as possible.
Fresh thinking to accelerate progress
People with type 1 diabetes are already benefiting from the cross-pollination of ideas between scientific fields. For instance, modern insulin pumps stem from technology originally invented by NASA scientists who were trying to solve the problem of monitoring astronauts’ health as missions sent them further and further into space.
The ambition and boldness of the Grand Challenge has given researchers from across scientific disciplines the opportunity to apply their skills and experience to type 1 diabetes.
Professor Dame Molly Stevens from University of Oxford, is an expert in biomaterials. She explores how materials interact with molecules, cells and tissues in the body and uses this knowledge to design advanced therapeutics and biosensing. In her Grand Challenge project, Professor Stevens is teaming up with stem cell researchers at King’s College London to find innovative ways to improve beta cell replacement strategies. For instance, by developing a protective ‘cage’ for transplanted beta cells.
“I’ve found an incredible synergy with my collaborators, allowing us to leverage the materials designed in my lab to improve the delivery of cell-based therapies for type 1 diabetes. It’s an exciting opportunity to make a real impact in this field! The Grand Challenge’s focus on high-risk, high-reward approaches really aligns with my passion for leveraging cutting-edge science to tackle complex medical challenges.”
Professor Shuomo Bhattacharya’s career has taken him from India where he researched rheumatic fever during his medical training, to studying heart development at Oxford University. But in recent years he’s been thinking about ticks, how they avoid being noticed by the body’s immune system, and how we could harness this system to combat autoimmune diseases.
“[The Grand Challenge] gave me the opportunity to apply stuff that I had discovered in a completely new arena that I had never even thought about. Most people including researchers and funding organisations tend to work in ‘silos’. Including diverse scientific backgrounds in a programme to tackle type 1 diabetes is the first step to break down these silos and bring in fresh ideas. I think the Grand Challenge got this right.”
Dr Tom Piers works with microglia – the immune cells of the brain. His research in this area has ranged from investigations into multiple sclerosis to Alzheimer’s disease progression. In recent years, Dr Piers has become interested applying his skills in diabetes and has been looking for a chance to do so.
“After my colleague Dr Criag Beall outlined the project idea, I have been very excited about the type of science that we can produce, crossing neuroscience, diabetes and cutting-edge biotechnology. But there are very limited funding opportunities for such high-risk high reward research. The Grand Challenge is addressing this and providing unprecedented support for these types of studies.”
Professor Eoin McKinney has primarily focused on immune responses to infections in our bodies. Together with the Grand Challenge, he’s now looking for new drugs that could potentially help prevent type 1 diabetes.
“The nature of research is that we think hard about a problem and become experts in applying particular methods to help solve it. But we also know that big leaps forward often come from applying our methods to a new problem or thinking differently by working with experts in other fields. It’s surprisingly difficult to do this – a side-effect of being focussed on one thing. That’s what excited us about the Grand Challenge.
We are being challenged to think differently and come up with more radical proposals to change the way things are done. This is where science can be most exciting and, hopefully, most productive too.”
A unique opportunity to change lives
Like Professor Bhattacharya who “spent most of that Christmas reading up about type 1 diabetes”, researchers from diverse fields of science are now excited about applying their knowledge and expertise to help people with type 1 diabetes.
Dr Piers, whose wife has type 1, sums it up:
“In my view, working across disciplines and disease areas with a diverse array of researchers is an excellent way of generating new, innovative and paradigm shifting ideas. I think we’re on the cusp of developing some very exciting new technologies to support people with type 1 and a scheme like the Grand Challenge will speed up their impact.”
You may also be interested in
Despite what you learned at school, insulin isn’t just made in the pancreas
June 16, 2025
This article is written by Dr Craig Beall and is republished from The Conversation under a Creative Commons license. Read the original article.

Dr Craig Beall in his research lab
Your brain makes insulin – the same insulin produced by your pancreas. The same insulin that is not produced in people with type 1 diabetes and the same insulin that does not work properly in people with type 2 diabetes.
Scientists have known for over 100 years about insulin producing cells in the pancreas. These spherical islands of cells, called islets, contain insulin producing beta cells.
But we’ve only just started to learn about brain insulin production. The fact that insulin is made there is still largely unknown, even among diabetes scientists, doctors and people with diabetes.
Yet, it was discovered there in the late 1970s – then promptly disregarded.
A study published in 1978 showed the levels of insulin in the rat brain were “at least 10 times higher than that found in plasma … and in some regions … 100 times higher”. If true, why isn’t this more widely known.
Because soon after this discovery, clear evidence showed the transfer of insulin from blood to brain. One study in 1983 measuring insulin in rodent brain said that “insulin found in these extracts was ultimately derived from pancreatic insulin”. They could not find the machinery to process insulin in the brain, at least with the tools available at the time.
This led to the assumption – for nearly the next 30 years – that all brain insulin came from the pancreas.
Insulin can and does move from the blood to the brain. But local sources of insulin are produced in specific places to do specific things.
The brain cells that make insulin
First, what is surprising about brain insulin production is that there is not one but at least six types of insulin-producing brain cell. Some have been confirmed in both rodent and human brain, others currently just in rodents.
One of the first brain cells shown to make insulin is the neurogliaform cell. These live in a brain area important for learning and memory. Most surprisingly, the production of insulin here depends on the amount of glucose present – a feature shared with pancreatic beta cells.
Its not clear what this insulin source does. Based on the location, it may contribute to cognitive function.
This area also has cells that create new neurons throughout life, called “neural progenitors”. These cells also make insulin.
A similar cell from the olfactory bulb, the processing centre for smell, also has insulin-producing progenitors. What insulin does here is still unknown.
But one insulin producing brain cell might regulate growth. A 2020 study showed that insulin is made and released from stress-sensing neurons in the mouse hypothalamus. This is a brain area that controls growth and metabolism. It also has the highest insulin levels in the human brain.
The researchers showed that stressing mice caused hypothalamic insulin production to decrease. This led to poorer growth in the animals. In the case of mice, their bodies were shorter.
Hypothalamic insulin maintained growth hormone levels in the pituitary gland. This is sometimes called the master gland as its involved in making or controlling production of other hormones. Having less local insulin meant less growth hormone production.
Then there is the choroid plexus. This is the brain region that makes cerebrospinal fluid. In humans, that is about half a litre of this clear colourless liquid every day.
Cells lining the choroid plexus – the epithelial cells – make a nourishing broth of growth factors and nutrients to keep the brain healthy. Only recently was insulin production found here in mice.
The choroid plexus secretes fluid directly into brain ventricles, the spaces deep inside the brain. This fluid flows around the whole brain, perhaps delivering insulin more widely.
One place it does travel to is the appetite control centre in the hypothalamus.
A 2023 study in mice showed that genetic control of insulin production by the choroid plexus could change food intake. The hypothalamus was rewired by changing choroid plexus insulin levels. Insulin released from here suppressed appetite.
Another source of insulin in the brain also reduces food intake. A 2022 found that insulin producing neurons at the back of the brain, called the hindbrain, reduced food intake in mice.
Might help the brain stay healthy as we age
So if brain insulin can change appetite, does it control blood sugar?
No. At least there is no evidence for this currently. It is unlikely this insulin leaves the brain. Therefore, its unlikely to control glucose levels in the same way.
Instead, insulin in the brain might help the brain stay healthy as we age. For example, Alzheimer’s disease is often, unofficially, termed type 3 diabetes. This is because the brain is insulin resistant in Alzheimer’s. It cannot properly use glucose either.
This is a big problem. Glucose is the main fuel for the brain. In fact, estimates suggest there is a 20% energy gap in Alzheimer’s. Even without brain cell loss, this alone will impair cognitive performance.
This has led to attempts to boost brain insulin. Spraying insulin into the nose can improve cognitive performance in Alzheimer’s, in some, but not all studies.
Brain glucose use also decreases over time and intranasal insulin also seems to limit this decrease.
Therefore, is more brain insulin always a good thing?
Not necessarily. In women specifically, higher levels of insulin in cerebrospinal fluid is associated with poorer cognitive performance.
There is still much to learn about brain insulin production. For example, which insulin source came first? The brain or the beta cell? Hopefully it doesn’t take another 30 years to find out.
But given the strength of evidence of brain insulin production, it won’t be long until our school textbooks are updated.
Read more about the Type 1 Diabetes Grand Challenge project led by Dr Craig Beall and Dr Thomas Piers that looks at how to make use of brain insulin production to improve beta cell therapies.
Viral triggers, ticks to tackle the immune system and a smart coat for insulin: Latest type 1 diabetes research
May 27, 2025
At the Type 1 Diabetes Grand Challenge, we support cutting-edge research aimed at bringing us closer to better treatments, and potentially, a cure. The research teams we fund are leaders in the field and their latest discoveries, highlighted here, are contributing to global efforts to transform the treatment of type 1 diabetes.
Connection between a common virus and type 1 diabetes
The development of type 1 diabetes is caused by a complex interplay of genetic and environmental factors, which trigger the immune system to turn on and destroy insulin-producing beta cells. The Type 1 Diabetes Grand Challenge is investing in research to learn more about precisely what goes wrong and how this process unfolds.
An international research team, including our Senior Research Fellow Professor Sarah Richardson, has just published the strongest evidence to date that enteroviruses play a role in the development of type 1 diabetes.
They studied pancreas samples from nearly 200 donors to check for signs of enterovirus infections. These included samples from people with and without type 1 diabetes, as well as individuals who had type 1 diabetes autoantibodies – early immune warning signals that indicate a high risk of developing the condition in the future.
They reported that signs of enterovirus infection were most common in people with type 1 diabetes who still had some functioning beta cells. Infections were also present in people with autoantibodies, but much less common in those without diabetes or in people with type 1 whose beta cells had already been completely destroyed.
A key finding was the presence of a protein made by the enterovirus, called VP1. The researchers found VP1 was strongly linked to high levels of HLA class I molecules. These are markers of an early immune system response in the pancreas. This is the first time these viral and immune markers have been found together – including in the early stages of type 1 before a diagnosis.
These findings offer the strongest evidence to date that enteroviruses may trigger or accelerate the onset of beta cell destruction in early type 1 diabetes. A clearer understanding of their role could lead to new approaches that target the immune processes they influence — potentially slowing or even preventing type 1.
‘Ticking’ beta cell protection
One part of a solution to curing type 1 diabetes is to transplant beta cells, either donated or grown in labs, into people with type 1 so they could produce their own insulin again. However, the immune system is primed to attack the new cells too. This happens partly because the transplanted cells release chemokines—chemical signals that attract immune cells. Blocking these signals is difficult, as there are many different types.
Scientists have discovered that ticks produce proteins called evasins that can block several of these signals at once. Professor Shoumo Bhattacharya’s team has studied these evasin proteins and identified the specific peptides that have this powerful blocking ability. They’ve established a new method that allows them to test many versions of these peptides rapidly, and pinpointed those that work best at calming the immune response.
Published in Communications Biology, these innovative findings lay the groundwork for Professors Shoumo Bhattacharya and David Hodson’s Grand Challenge project. They aim to use these peptides to help protect new beta cells from immune attack after being transplanted. This could create a safer alternative to immunosuppressive drugs and improve the success of beta cell therapies for type 1 diabetes.
Giving insulin a smart coat
Professor Zhen Gu’s team at Zhejiang University in China has developed a new insulin delivery system, called i-crystal, as reported in their latest publication in Nature Nanotechnology.
Designed to help people with type 1 diabetes keep blood sugar levels within target range, the system uses specially coated insulin crystals that release insulin slowly and steadily. The coating is a unique membrane that enables the crystals to respond precisely to changes in blood sugar and ketone levels. This means insulin is only released when blood sugar levels rise and stops when it drops, helping people with type 1 avoid highs (hypers) and lows (hypos).
Early tests show that i-crystal could help regulate blood sugar levels for over a month in mice after just one injection, and for more than three weeks in minipigs after five daily doses. This suggests this delivery system could reduce the need for frequent injections and make daily diabetes management easier.
If successful, i-crystal could offer a safer, longer-lasting, and more adaptable form of insulin therapy for people living with type 1 diabetes.